MaxCyte, Inc. announced a strategic collaboration with Washington University in St. Louis, a leading institution in acute myeloid leukemia (AML) research. MaxCyte and the university will develop unique immunotherapy drug candidates, based on MaxCyte's proprietary cell engineering platform technology, CARMA.
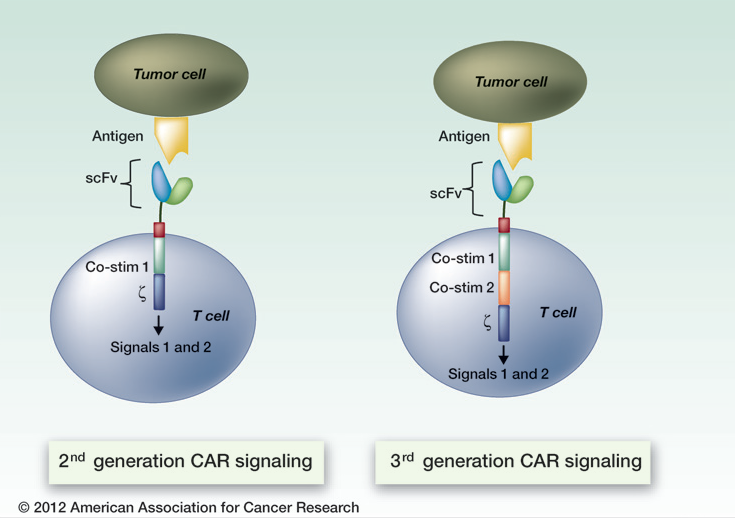
CARMA allows simple and rapid manufacture of advanced cancer treatments that utilize a patient's own immune system and is differentiated from traditional CAR therapy due to its use of mRNA to engineer immune cells delivered back into a patient. By utilizing transient expression via mRNA delivery, CARMA allows control over severe adverse effects, opening the high potency of CAR immunotherapies to a broader range of cancers than traditional CAR approaches.
"We're excited to initiate this collaboration with Washington University in St. Louis, one of the leading medical research centers worldwide, to develop a new generation of CAR therapy for blood cancers using MaxCyte's CARMA therapeutic platform," said Doug Doerfler, president & CEO of MaxCyte, Inc. "This collaboration is a natural extension of our efforts in this new class of therapies, given our previously announced collaboration with the Johns Hopkins Kimmel Cancer Center for pre-clinical development of CAR therapy for solid cancers. Our CARMA platform shows great promise for developing therapies for patients with both solid and liquid tumor types. It provides rapid, cost-effective manufacturing and delivery of cellular therapies to patients without many of the toxicities associated with other CAR-based approaches."
John DiPersio, M.D., Ph.D., chief of the Division of Oncology at Washington University School of Medicine and Deputy Director of the Siteman Cancer Center, and members of his team will collaborate with MaxCyte to conduct preclinical research with a focus on developing a potential investigational CAR therapy targeting acute myeloid leukemia and other related blood cancers. Financial terms were not disclosed.
The preclinical studies that the two collaborators anticipate as a result of this agreement will take advantage of the rapid, low-cost therapeutic development and manufacturing process enabled by the MaxCyte technology. As such, the financial commitment in the program would not be significant at this stage. ?
Researchers are investigating the use of CARMA, the company's patented approach to CAR, to generate the next class of immunotherapy for cancer, aiming to improve on patient outcomes for those with either solid or liquid tumor types. CARMA-engineered immune cells seek and destroy cancer cells with the potential to deliver precise therapies for patients against a range of cancers, without the cost and complexity of centralized manufacturing and adverse effects seen in first-generation, viral-based CAR therapies. MaxCyte believes that the promising preclinical results obtained from the collaboration with The Johns Hopkins Kimmel Cancer Center, along with further studies, will result in an investigational new drug (IND) filing with the US Food and Drug Administration (FDA) in 2017 for patients with ovarian cancer. The company continues to explore new targets and additional collaborators to advance the CARMA platform.
Maxcyte, washington university collaborate, develop next generation car based cell therapies